Publication in Journal of the American Chemical Society (2026)
20 02 2026
A scientific paper by the research group of Prof. Krzysztof Woźniak has been published in the Journal of the American Chemical Society. The study concerns the redistribution of electron density in calcite prior to the high-pressure phase transition from Calcite I to Calcite II.
The co-authors of the paper are Dr. Marcin Stachowicz from the Faculty of Geology, Ms. Agnieszka Huć – a PhD student of Prof. Woźniak at the Faculty of Geology, Tomasz Poręba from the ESRF (Grenoble, France) and ETH Lausanne (Switzerland), Mohamed Mezouar from the ESRF, and Przemysław Dera from the University of Hawaiʻi at Mānoa (Honolulu).
Understanding the fate of carbonate rocks at mantle depths is crucial for modelling the global carbon cycle. Calcite, CaCO3, undergoes a pressure-induced transition from calcite-I (R3̅c) to calcite-II (P21/c) close to 1.6 GPa, but the effects of these changes on chemical reactivity and electronic properties remain poorly understood. Combining high-pressure X-ray diffraction at the ID27 beamline of the EBS-ESRF synchrotron with charge density analysis reveals subtle electronic and structural changes in the calcite structure with pressure. By determining the integrated charges, atomic basin volumes, and shapes of Ca, C, and O atoms, we effectively decompose the equation of state into individual atomic contributions. The analysis reveals that the phase transition is preceded by a discontinuous charge redistribution between Ca, C, and O atoms, occurring before the symmetry change and reversing upon decompression without hysteresis. Notably, carbon displays negative atomic compressibility, expanding under pressure due to electron uptake, while Ca and O atoms behave conventionally. Our findings demonstrate that experimental charge density methods can resolve pressure-induced electron density rearrangements with unprecedented detail, offering new perspectives on the mineralogy of Earth’s interior.
Our results show that experimental charge-density analysis methods make it possible to capture pressure-induced rearrangements of electron density with unprecedented accuracy and precision, opening new perspectives in the study of the Earth’s interior mineralogy.
M. Stachowicz*, A. Huć, T. Poręba, M. Mezouar, P. Dera, K. Woźniak*, Charge Shift in Calcite before High-Pressure Phase Transition, J. Am. Chem. Soc. (2026) Published on-line Feb.13, 2026, https://doi.org/10.1021/jacs.5c12911
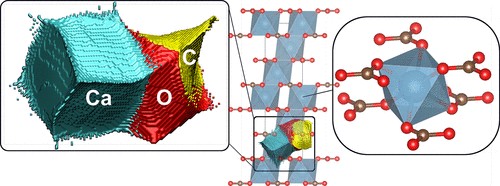
Figure 1. Atomic topology and coordination environment in the calcite-I crystal structure. Left: Bader’s quantum theory of atoms in molecules and crystals (QTAMC) representation of an asymmetric unit, showing atomic basins of calcium (blue), oxygen (red), and carbon (yellow). Center: Crystal structure of calcite-I, illustrating planar layers of CO32– groups perpendicular to the [001] axis. Right: Calcium coordination polyhedron, with six oxygen atoms (red spheres) from six distinct carbonate groups with Ca in linking adjacent carbonate sheets.
